Despite decades of progress, cancer care has followed the same pattern: doctors step in only after the disease has already taken root. By that point, cancer cells have learned how to survive, evade the immune system, and sometimes spread to other parts of the body. Preventing cancer in the first place would generally be easier than treating an established one. A new recombinant protein vaccine approach seeks to shift this paradigm by training the immune system to recognise α-lactalbumin, a protein expressed in many cases of triple-negative breast cancer (TNBC). Early clinical testing has reported encouraging results with this strategy, offering hope for preventing one of the most aggressive and hard-to-treat types of breast cancer.
The Triple-Negative Breast Cancer (TNBC) Threat
In 2021, Chase Johnson was 31 when she noticed something odd. Her dog had grown unusually restless and kept clinging to her. One day, the dog pressed his nose firmly against her breast. Johnson felt a hard lump and knew something was wrong. She then went to see a doctor and was unfortunately diagnosed with triple-negative breast cancer (TNBC), a type of breast cancer that often affects younger women and behaves more aggressively. After months of intensive treatment with intravenous chemotherapy and surgery, followed by additional oral chemotherapy and radiotherapy, Johnson was declared cancer-free.
Had this been a more common hormone-responsive breast cancer, the long-term outlook would have been more reassuring for Johnson. But TNBC carries a different risk profile. It is several times more likely to lead to metastatic recurrence. Even after successful treatment, around 40% of patients experience relapse within five years. When this happens, the relapse is frequently metastatic, involving distant organs such as the brain, lungs or liver – sites where therapeutic options are fewer and clinical outcomes are poorer. Compounding this uncertainty, no standardised clinical test can reliably predict recurrence risk in TNBC survivors at the moment.
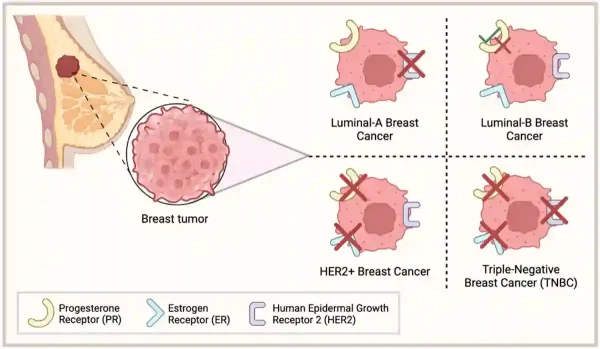
Worse still, TNBC accounts for about 15% of all breast cancer cases and is notoriously difficult to treat. By definition, ‘triple-negative’ in TNBC means it lacks three molecular targets that underpin most modern breast cancer therapies: the oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 (HER2) (Figure 1). Without these targets, standard hormonal therapies and HER2-targeting drugs are ineffective. Chemotherapy remains the main option, but it does little to prevent the cancer from coming back.
For patients like Johnson, who have already endured intensive therapies and even been declared cancer-free, the lingering risk of recurrence creates a prolonged period of uncertainty. It was this persistent risk that led Johnson to enrol in an early-phase clinical trial testing a novel recombinant protein vaccine strategy for TNBC. The vaccine is designed to train the immune system to recognise α-lactalbumin, a protein abnormally expressed in many TNBC cells. By doing so, the vaccine aims to prevent recurrence and, in high-risk individuals, to prevent TNBC from developing altogether.
Figure 1. How breast cancers are classified by surface markers. Breast cancers are grouped by whether their cells express oestrogen receptors, progesterone receptors and/or human epidermal growth receptor 2 (HER2). Luminal cancers keep one or more hormone receptors, HER2-positive cancers rely on the HER2 switch, and triple-negative breast cancer (TNBC) lacks all three receptors. Source: Kirkby et al. (2023), Cancers.
α-Lactalbumin Vaccine for TNBC: The Rationale
What makes this triple-negative breast cancer (TNBC) vaccine concept so unusual is its target. Rather than aiming at an infectious agent, the vaccine uses a laboratory-produced (recombinant) version of α-lactalbumin to safely expose the immune system to this protein and prime a targeted response. α-Lactalbumin is a milk protein normally active only during late pregnancy and breastfeeding, and otherwise silent in adult breast tissue. Yet in over 70% of TNBC tumours, α-lactalbumin is abnormally re-expressed, effectively flagging cancer cells in a way the vaccine-trained immune system can recognise.
This is the rationale behind what pioneering investigators at the Cleveland Clinic call the retired protein hypothesis. Some proteins do important work early in life, such as during breastfeeding, then effectively retire when they are no longer needed. When a tumour later reactivates these retired proteins, they become a rare kind of target: self-derived proteins that are not actively used in non-cancerous cells. This markedly lowers the chance that an immune attack aimed at the tumour will spill over and damage healthy tissue (i.e., autoimmunity).
While the precise reason why TNBC reactivates α-lactalbumin remains unclear, scientists hypothesise that the absence of oestrogen and progesterone receptors in TNBC disrupts hormonal signalling pathways that normally silence α-lactalbumin. Hence, this phenomenon has created a window of vulnerability that vaccine designers aim to target. “Tumours do bizarre things,” said Vincent Tuohy, PhD, an immunologist at Lerner Research Institute, Cleveland Clinic, U.S., who invented the α-lactalbumin vaccine. “Expressing α-lactalbumin is a mistake that the TNBC tumours make, and we’re taking advantage of this mistake.”
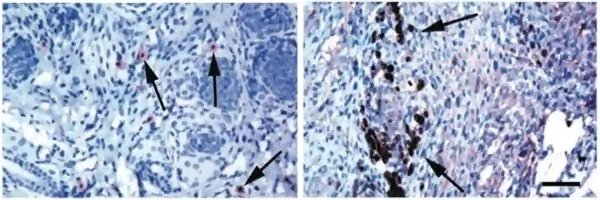
In preclinical mouse models, α-lactalbumin vaccination protected against breast tumour without triggering inflammation in non-lactating mice. In contrast, vaccinating lactating mice led to breast inflammation and lactation failure (Figure 2). This trade-off is exactly what you would predict if α-lactalbumin is only expressed during late pregnancy or breastfeeding. Therefore, unless future studies clarify otherwise, the α-lactalbumin vaccine is intended for individuals who do not plan to become pregnant or breastfeed. As scientists at the Cleveland Clinic wrote, “α-lactalbumin vaccination may provide safe and effective protection against the development of breast cancer for women in their post-childbearing, premenopausal years, when lactation is readily avoidable and risk for developing breast cancer is high.”
Figure 2. How the immune system responds to the α-lactalbumin vaccine depending on lactation status. These microscope images show breast tissue from vaccinated mice. Left panel: In non-lactating mice, only a few immune T cells (arrows) are seen patrolling the breast tissue, with no signs of inflammation. Right panel: In lactating mice, where α-lactalbumin is naturally switched on, the same vaccination triggers a strong immune reaction, with large numbers of immune cells clustering within the breast tissue (dark-stained areas). Source: Jaini et al. (2010), Nature Medicine.
α-Lactalbumin Vaccine for TNBC: Early Clinical Results
The recombinant α-lactalbumin vaccine has now advanced from preclinical models into early human testing. In the Phase 1 clinical trial, participants received the recombinant α-lactalbumin vaccine formulated with zymosan (i.e., a yeast-derived adjuvant that helps activate the immune response). The vaccine was administered as three injections given at two-week intervals, using a standard dose-escalation design. To assess whether the vaccine successfully engaged the immune system, investigators measured α-lactalbumin-specific immune responses, including T-cell activation, cytokine production and antibody responses.
As with all first-in-human studies, the goal of this Phase 1 trial was deliberately limited. It was not designed to show that the vaccine prevents cancer recurrence or the formation of new tumours. Instead, it sought to answer two foundational questions:
- Is the vaccine safe and tolerable?
- Can it reliably induce an immune response against α-lactalbumin in humans?
To address these questions, the trial enrolled 35 participants across three clinical groups:
- Phase Ia included triple-negative breast cancer (TNBC) survivors who had completed standard therapy and were cancer-free but at high risk of recurrence.
- Phase Ib included cancer-free individuals with a strong genetic predisposition to TNBC, such as BRCA mutation carriers, who were undergoing risk-reducing surgery.
- Phase Ic included patients with residual TNBC after chemo-immunotherapy and surgery who were receiving adjuvant immunotherapy.
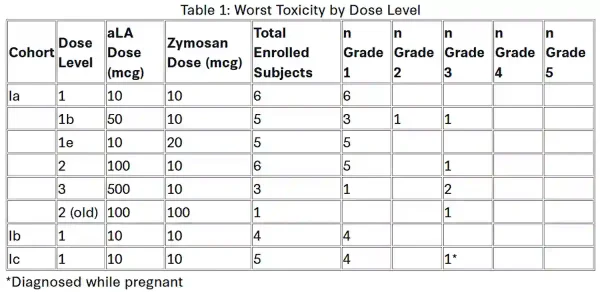
Overall, the vaccine was safe and well-tolerated. About 74% of participants developed a measurable immune response to α-lactalbumin. Most side effects were mild and limited to the injection site, like redness or discomfort. As is typical in dose-escalation studies, more severe local reactions, such as ulceration requiring drainage, occurred at higher doses and helped set the maximum tolerated dose. This procedure is a standard part of early clinical trials (Figure 3). As principal investigator, G. Thomas Budd, M.D., of the Cleveland Clinic’s Taussig Cancer Institute noted, “The results from this trial are promising, as they suggest the investigational vaccine is not only safe and well tolerated but also capable of inducing immune responses in over 70% of participants.”
Figure 3. Safety profile of the α-lactalbumin vaccine at different dose levels across the three study cohorts (Phase Ia to Ic). Side effects were graded from Grade 1 (mild) to Grade 5 (life-threatening or fatal). Most participants experienced only mild (Grade 1) reactions, typically at the injection site. More severe Grade 2-3 reactions (i.e., ulceration requiring drainage) were uncommon and helped investigators determine the maximum tolerated dose of the vaccine. Source: Johnson et al. (2025), Poster Session 4 at San Antonio Breast Cancer Symposium.
However, demonstrating immunogenicity alone does not yet mean the vaccine prevents cancer. Rather, it establishes a critical prerequisite: that α-lactalbumin-specific immunity can be safely generated in humans at appropriate doses. This milestone paves the way for the Phase 2 trial to test whether such immune priming translates into real clinical benefits.
What is encouraging is that this vaccine strategy is being tested in a context where cancer vaccines are most likely to succeed. Unlike established tumours, newly emerging cancer cells have had little time to evolve immune-escape mechanisms, making them more vulnerable to immune surveillance. If future trials demonstrate that α-lactalbumin-targeted immunity can reduce recurrence or prevent tumour formation in high-risk individuals, it would mark a fundamental shift in how cancer is approached: not as a disease to be treated once established, but as a biological process that can be intercepted before it takes hold.
“There was a time when we would say HER2 is the worst type of breast cancer you can have, then along came HER2-targeting therapies and now all of the sudden one of the worst prognosis markers becomes one of the best,” said Larry Norton, M.D., medical director of the Evelyn H. Lauder Breast Centre in New York, who is impressed with the early trial results. “This could be the story of triple-negative breast cancer if we find a target for it.”
Even if the recombinant α-lactalbumin vaccine ultimately fails to demonstrate clinical benefit in Phase 2 testing, the effort itself remains consequential. As Dr Norton has noted, advances in tumour biology are steadily improving our ability to identify unusual molecules on cancer cells that can be targeted therapeutically. In that sense, α-lactalbumin may not be the only solution, but the first of several vulnerabilities in TNBC that could be targeted.